WARNING: This product is for research use only, not for human or veterinary use.
Hodoodo CAT#: H577705
CAS#: 192927-63-2 (HCl hydrate)
Description: Moxifloxacin hydrochloride monohydrate is a fourth-generation synthetic fluoroquinolone antibacterial agent.
Hodoodo Cat#: H577705
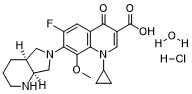
Name: Moxifloxacin hydrochloride monohydrate
CAS#: 192927-63-2 (HCl hydrate)
Chemical Formula: C21H27ClFN3O5
Exact Mass: 455.16
Molecular Weight: 455.910
Elemental Analysis: C, 55.32; H, 5.97; Cl, 7.78; F, 4.17; N, 9.22; O, 17.55
Related CAS #: 151096-09-2 (free base) 186826-86-8 (HCl) 192927-63-2 (HCl hydrate)
Synonym: Moxifloxacin hydrochloride monohydrate; Moxifloxacin HCl monohydrate
IUPAC/Chemical Name: 7-((4aS,7aS)-Octahydro-1h-pyrrolo(3,4-b)pyridin-6-yl)-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid hydrate hydrochloride
InChi Key: SKZIMSDWAIZNDD-WJMOHVQJSA-N
InChi Code: InChI=1S/C21H24FN3O4.ClH.H2O/c1-29-20-17-13(19(26)14(21(27)28)9-25(17)12-4-5-12)7-15(22)18(20)24-8-11-3-2-6-23-16(11)10-24;;/h7,9,11-12,16,23H,2-6,8,10H2,1H3,(H,27,28);1H;1H2/t11-,16+;;/m0../s1
SMILES Code: O.Cl.COc1c(N2C[C@@H]3CCCN[C@@H]3C2)c(F)cc4C(=O)C(=CN(C5CC5)c14)C(=O)O
Appearance: Solid powder
Purity: >98% (or refer to the Certificate of Analysis)
Shipping Condition: Shipped under ambient temperature as non-hazardous chemical. This product is stable enough for a few weeks during ordinary shipping and time spent in Customs.
Storage Condition: Dry, dark and at 0 - 4 C for short term (days to weeks) or -20 C for long term (months to years).
Solubility: To be determined
Shelf Life: >2 years if stored properly
Drug Formulation: To be determined
Stock Solution Storage: 0 - 4 C for short term (days to weeks), or -20 C for long term (months).
HS Tariff Code: 2934.99.9001
More Info:
| Biological target: | |
| In vitro activity: | |
| In vivo activity: |
The following data is based on the product molecular weight 455.91 Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which will affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 1.15 mL | 5.76 mL | 11.51 mL |
| 5 mM | 0.23 mL | 1.15 mL | 2.3 mL |
| 10 mM | 0.12 mL | 0.58 mL | 1.15 mL |
| 50 mM | 0.02 mL | 0.12 mL | 0.23 mL |
| Formulation protocol: | |
| In vitro protocol: | |
| In vivo protocol: |
1: Gillespie SH. The role of moxifloxacin in tuberculosis therapy. Eur Respir Rev. 2016 Mar;25(139):19-28. doi: 10.1183/16000617.0085-2015. Review. PubMed PMID: 26929417.
2: Chen Z, Liang JQ, Wang JH, Feng SS, Zhang GY. Moxifloxacin plus standard first-line therapy in the treatment of pulmonary tuberculosis: A meta-analysis. Tuberculosis (Edinb). 2015 Jul;95(4):490-6. doi: 10.1016/j.tube.2015.03.014. Epub 2015 Apr 16. Review. PubMed PMID: 25964137.
3: Ovchinnikov AY, Edzhe MA, Miroshnichenko NA, Hon EM, Korostelev SA. [15-year experience of moxifloxacin in the treatment of patients with bacterial rhinosinusitis]. Vestn Otorinolaringol. 2015;80(3):75-9. Review. Russian. PubMed PMID: 26331163.
4: Chilet-Rosell E, Ruiz-Cantero MT, Pardo MA. Gender analysis of moxifloxacin clinical trials. J Womens Health (Larchmt). 2014 Jan;23(1):77-104. doi: 10.1089/jwh.2012.4171. Epub 2013 Nov 1. Review. PubMed PMID: 24180298; PubMed Central PMCID: PMC3881443.
5: Linertová R, Abreu-González R, García-Pérez L, Alonso-Plasencia M, Cordovés-Dorta LM, Abreu-Reyes JA, Serrano-Aguilar P. Intracameral cefuroxime and moxifloxacin used as endophthalmitis prophylaxis after cataract surgery: systematic review of effectiveness and cost-effectiveness. Clin Ophthalmol. 2014 Aug 14;8:1515-22. doi: 10.2147/OPTH.S59776. eCollection 2014. Review. PubMed PMID: 25152613; PubMed Central PMCID: PMC4140229.